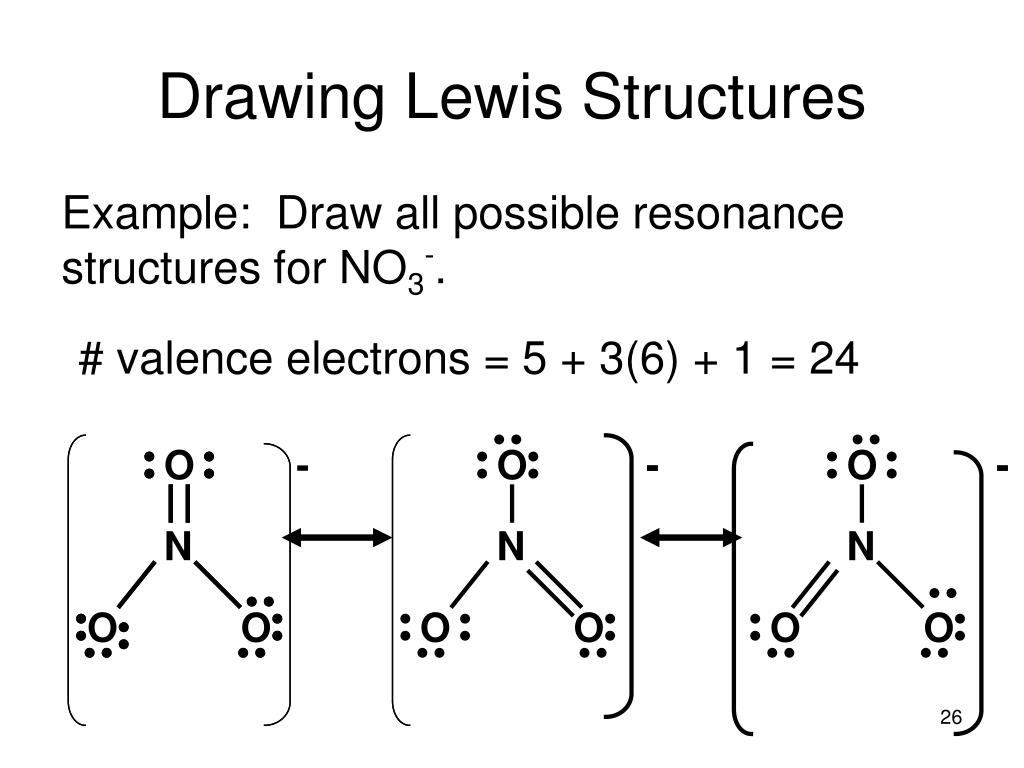
Drawing Lewis Structures Practice
Drawing Lewis Structures Practice - Web this online quiz is intended to give you extra practice in identifying and drawing lewis dot structures as well as predicting ion formation. Web this chemistry video provides a basic introduction into how to draw lewis structures of common molecules such as cl2, o2, of2, ch4, nh3, h2o, c2h2, and n2h4. Web practice drawing these lewis structures and don't worry we will go over all the answers step by step. You should consult the lewis structure rules and a periodic table while doing this exercise. See the following lewis dot structure diagrams for a few covalent compounds. Web the electrons in the outermost shell are called the _____ electrons. For the following molecules or ions (where the central atom is underlined): Write the correct skeletal structure for the molecule. Web in this video you’ll learn how to draw lewis dot structures for covalent compounds. The example is for the nitrate ion. This quiz aligns with the following ngss standard(s): Web draw a lewis structure for each of the following:b. Lewis diagram of the cyanide ion (cn⁻). Web practice drawing these lewis structures and don't worry we will go over all the answers step by step. Lewis diagram of the cyanide ion (cn⁻) One line is a single bond with 2 bonding electrons, two lines is a double bond with 4 bonding electrons, and three lines. Draw a lewis structure for each of the following:c. Web the skeletal structure of ethanethiol is shown below. Web write the lewis structures for carbon tetrachloride and phosgene. Then name their electron arrangement, shape, and bond angles. Web here are the steps to draw a lewis structure. The video covers the basic lewis structures you'll see in an introductor. Web practice drawing these lewis structures and don't worry we will go over all the answers step by step. Draw a lewis structure for each of the following:d. Change the following condensed structures to. The example is for the nitrate ion. Figure out how many electrons the molecule must have, based on the number of valence electrons in each. The video covers the basic lewis structures for a general chemistry class. Draw the lewis structure for the following molecules. Web here are the steps to draw a lewis structure. Be sure you know how to draw correct lewis dot structures and are able to correctly predict the electronic arrangement and molecular geometry before going on to the lab assignment. Here, we will be using the determined total number of valence electrons per atom and drawing them in the proper places. This quiz aligns with the following ngss standard(s): Ch. Lewis diagram of the cyanide ion (cn⁻) Lewis diagram of formaldehyde (ch₂o) worked example: Draw a lewis structure for each of the following:d. Web practise drawing the lewis structure of molecules using the exercises below. Which of the following correctly completes the lewis diagram of ethanethiol? The following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Which of the following correctly completes the lewis diagram of ethanethiol? Web this chemistry video provides a basic introduction into how to draw lewis structures of common molecules such as cl2, o2, of2, ch4, nh3, h2o, c2h2, and n2h4.. Ch 2 br 2 d. Web examples for drawing lewis structures for covalent bonds. Lewis diagram of the cyanide ion (cn⁻). Web here are the steps to draw a lewis structure. Reference the “how to draw a lewis dot structure” for a step by step guide. The following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Web this online quiz is intended to give you extra practice in identifying and drawing lewis dot structures as well as predicting ion formation. Web drawing lewis structures for molecules with one central atom: A lewis structure can be. See the following lewis dot structure diagrams for a few covalent compounds. This quiz aligns with the following ngss standard(s): A lewis structure is a diagram that shows the chemical bonds between atoms in a molecule and the valence electrons or lone pairs of electrons.the diagram is also called a lewis dot diagram, lewis dot formula, or electron dot diagram.. Web practice drawing lewis structures with answers and explanation. A lewis structure is a diagram that shows the chemical bonds between atoms in a molecule and the valence electrons or lone pairs of electrons.the diagram is also called a lewis dot diagram, lewis dot formula, or electron dot diagram. Figure out how many electrons the molecule must have, based on. Web examples for drawing lewis structures for covalent bonds. The video covers the basic lewis structures for a general chemistry class. Ch 2 br 2 d. Web in this video you’ll learn how to draw lewis dot structures for covalent compounds. This video will explain how to find the formal charges. Be sure you know how to draw correct lewis dot structures and are able to correctly predict the electronic arrangement and molecular geometry before going on to the lab assignment. Lewis diagram of the cyanide ion (cn⁻) Web practise drawing the lewis structure of molecules using the exercises below. Figure out how many electrons the molecule must have, based on the number of valence electrons in each. Write the correct skeletal structure for the molecule. Draw the lewis dot structures for each of the following molecules: See the following lewis dot structure diagrams for a few covalent compounds. A periodic table will be available for the exam, but the list of rules will not be available, so this is a chance to practice using the rules to help you remember them! Web this chemistry video provides a basic introduction into how to draw lewis structures of common molecules such as cl2, o2, of2, ch4, nh3, h2o, c2h2, and n2h4. Reference the “how to draw a lewis dot structure” for a step by step guide. Yes, covalent bonds come in pairs which are represented by lines in lewis structures. Then name their electron arrangement, shape, and bond angles. Draw the lewis dot structure for each of the following polyatomic ions: A lewis structure is a diagram that shows the chemical bonds between atoms in a molecule and the valence electrons or lone pairs of electrons.the diagram is also called a lewis dot diagram, lewis dot formula, or electron dot diagram. Web this online quiz is intended to give you extra practice in identifying and drawing lewis dot structures as well as predicting ion formation. Web sure if your structure is correct, do a formal charge check.How to draw Lewis Structures a step by step tutorial Middle School
A StepbyStep Guide on Drawing Lewis Structures Inflation Protection
How to Draw a Lewis Structure
How to Draw a Lewis structure Easy and Quick Method
How to Draw a Lewis Structure
CP Drawing Lewis Structures Practice YouTube
Sf2 Lewis Structure How To Draw The Lewis Structure For
Quick & Easy 5 Steps to Drawing Lewis Structures with Examples
Drawing Lewis Dot Structures for Chemistry dummies
3 Ways to Draw Lewis Dot Structures wikiHow
Web The Electrons In The Outermost Shell Are Called The _____ Electrons.
Web Examples For Drawing Lewis Structures For Covalent Bonds.
Here, We Will Be Using The Determined Total Number Of Valence Electrons Per Atom And Drawing Them In The Proper Places.
This Quiz Aligns With The Following Ngss Standard(S):
Related Post:

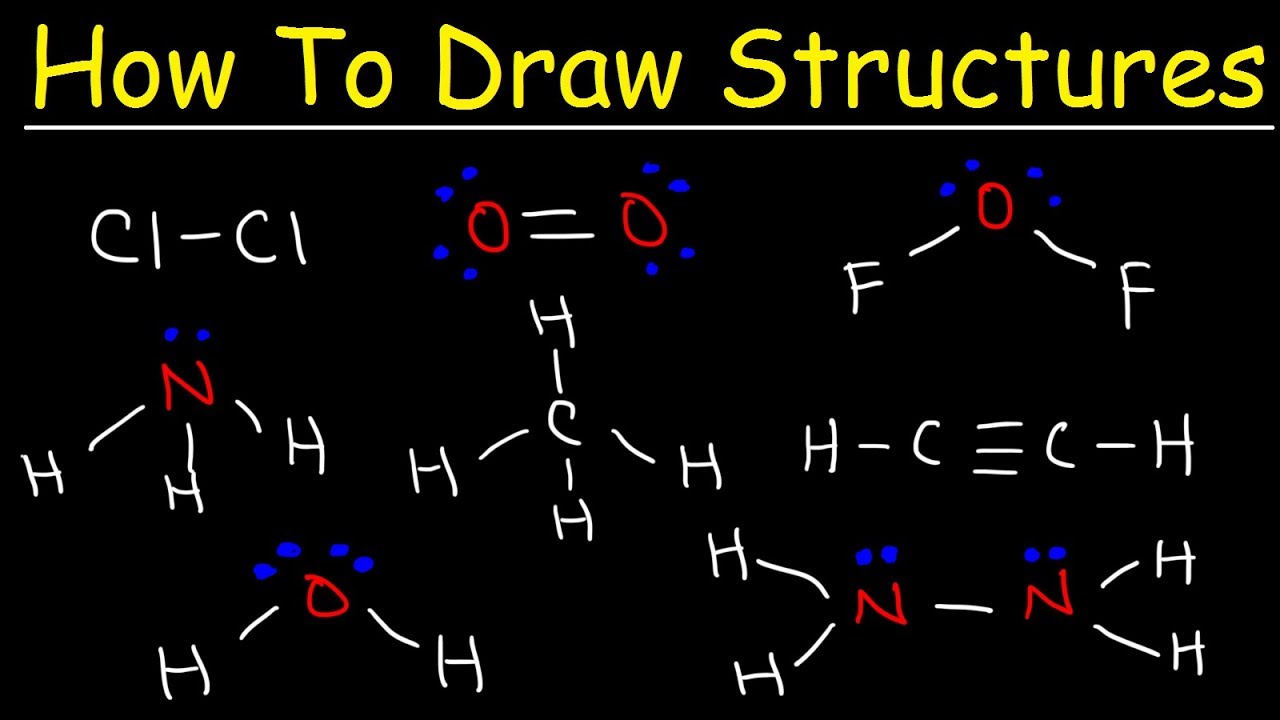
/Lewis-dot-structure-58e5390f3df78c5162b4c3db.jpg)